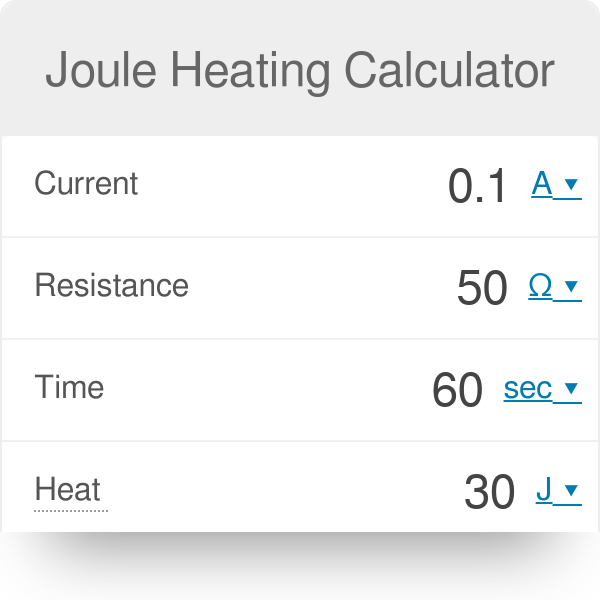
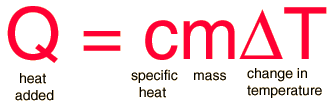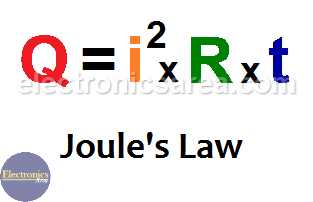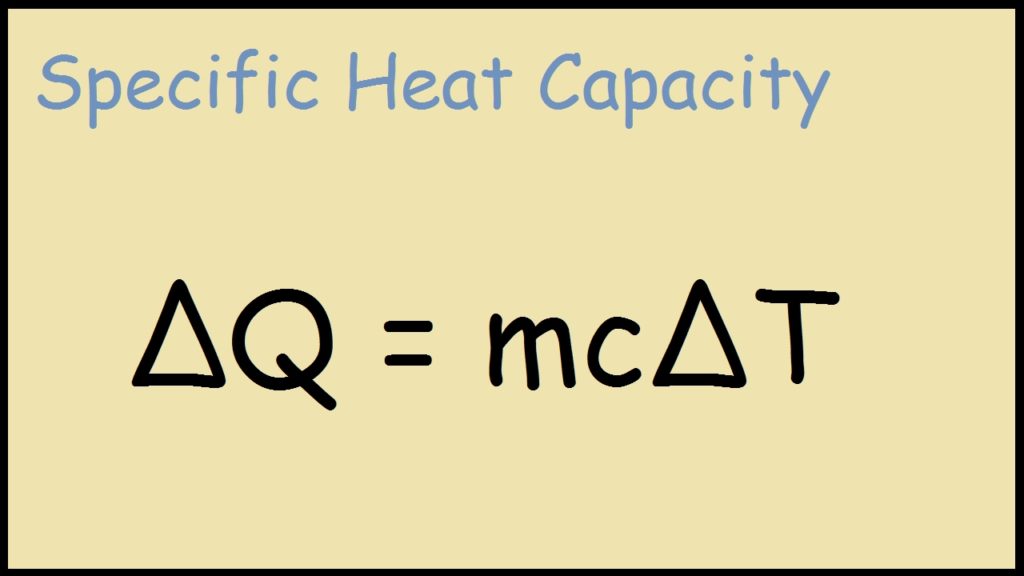Thermodynamics Ch 10 Energy Sections Thermodynamics The 1st Law of Thermodynamics The Law of Conservation of Energy is also known as The 1st. - ppt download
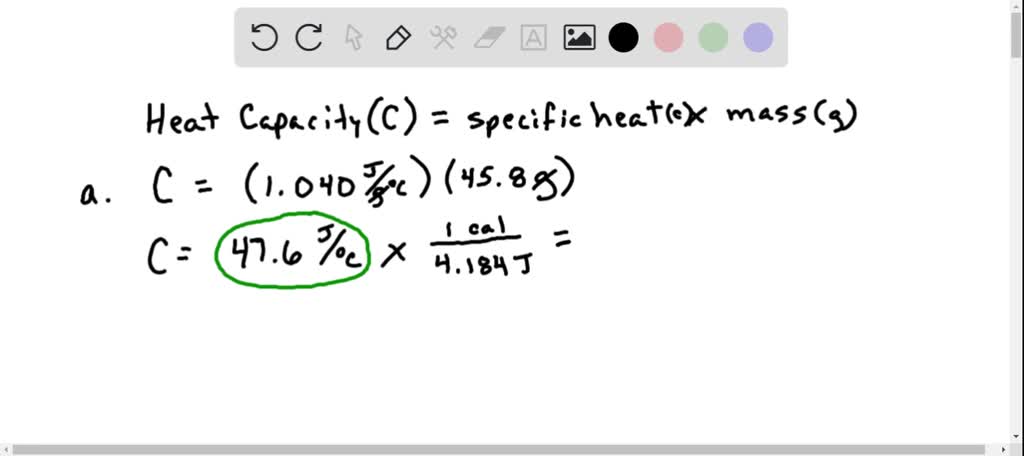
SOLVED:Calculate the heat capacity, in joules and in calories per degree, of the following: (a) 45.8 g of nitrogen gas (b) 1.00 pound of aluminum metal

Question Video: Calculating the Heat Energy Transferred to Water Using Its Specific Heat Capacity | Nagwa

Calculate the amount of heat (in Joules) required to convert 25 grams of water at 100 ^∘ C into steam?(Given : heat of vaporization of water = 2257 J/g)

If 294 joules of heat energy is required to raise the temperature of 2 moles of an ideal gas from 30^(@)C to 35^(@)C at constant pressure, then the specific heat at constant